About Seawater
Water
 Water is a liquid made up of H2O, a triangular molecule with slightly flexible arms between the larger oxygen atom at its center with the two smaller hydrogen molecules.
Water is a liquid made up of H2O, a triangular molecule with slightly flexible arms between the larger oxygen atom at its center with the two smaller hydrogen molecules.
Not only is this molecule oddly shaped, so that it is hard to pack water molecules together, but the efficiency of packing changes with temperature and pressure as these arms vibrate and deform.
Water can also freeze and become a solid, with a density less than that of water as the packing structure of ice has many voids, or evaporate and become a gas, which can require (or liberate) large amounts of heat energy.
Seawater
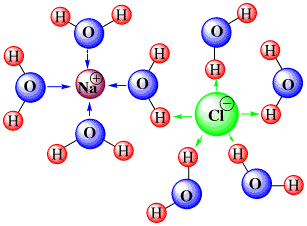 Seawater is water that includes dissolved matter. Rivers and lakes also contain dissolved matter, but typically a dissolved molecule or ion in freshwater is separated from other non-water molecules by dozens of water molecules. Since these non-water molecules are then too far apart to affect each other, freshwaters can often be treated as ‘dilute’ solutions, where the effects of dissolved molecules and electrolytes (salts, acids, and bases) are essentially independent of one another, making properties of the solution relatively easy to predict.
Seawater is water that includes dissolved matter. Rivers and lakes also contain dissolved matter, but typically a dissolved molecule or ion in freshwater is separated from other non-water molecules by dozens of water molecules. Since these non-water molecules are then too far apart to affect each other, freshwaters can often be treated as ‘dilute’ solutions, where the effects of dissolved molecules and electrolytes (salts, acids, and bases) are essentially independent of one another, making properties of the solution relatively easy to predict.
However, in seawater, these dissolved molecules are separated from each other by only about 4 water molecules on average. This means that the different non-water molecules can affect each other, making it vastly more difficult to predict what the bulk properties of seawater might be from first principles. This is particularly true for the electrically charged ions that result from the dissolution of sea salt.
These bulk properties can be physical (for example, the density or speed of sound in seawater), or chemical (affecting the way in which other soluble species such as trace metals, or those related to the carbonate system, combine and react), or electrical (affecting the way in which electricity can flow through it).
The role of standards
 What is the density of water at a particular temperature? What is the density of seawater at that temperature, pressure, and salinity? These values are needed for many purposes, and we don’t want to have to measure them every time they are needed. Hence the role of STANDARDS.
What is the density of water at a particular temperature? What is the density of seawater at that temperature, pressure, and salinity? These values are needed for many purposes, and we don’t want to have to measure them every time they are needed. Hence the role of STANDARDS.
Standards are embodied in tables, or equations, that summarize experimental measurements. They are written down in a document (the “standard” itself) which can then be used as a reference. When two or more scientists/engineers agree on this reference, then they are “talking the same language”.
But who makes standards, and who decides which ones are better for a particular purpose?
Standards in Seawater
Some of the more important standards related to seawater are:
- International Temperature Scale 1990 (ITS-90), replacing the older International Practical Temperature Scale 1968 (IPTS-68)
- Practical Salinity Scale 1978 (PSS-78), which governs conductance-based salinity determination.
- the Equation of State 1980 (EOS-80), a loosely related set of standards for the density and some other properties of seawater.
- The IAPWS Formulation 2008 for the Thermodynamic Properties of Seawater (IAPWS R13-08), which describes the bulk properties of IAPSO Standard Seawater using the Reference Composition Salinity Scale.
- The Thermodynamic Equation of Seawater 2010 (TEOS-10), the current best attempt to describe the bulk properties of seawater from anywhere in the world.
Maintaining the relationship between these interdependent standards, produced by different organizations at different times, is one of the tasks of JCS.
